A KRAS-Targeted mRNA Cancer Vaccines: Synergistic Mechanisms and Future Perspectives in Combination Immunotherapy
Keywords:
KRAS, mRNA vaccine, nucleic acid therapy, combination therapy, tumor immune microenvironment, resistance mechanismsAbstract
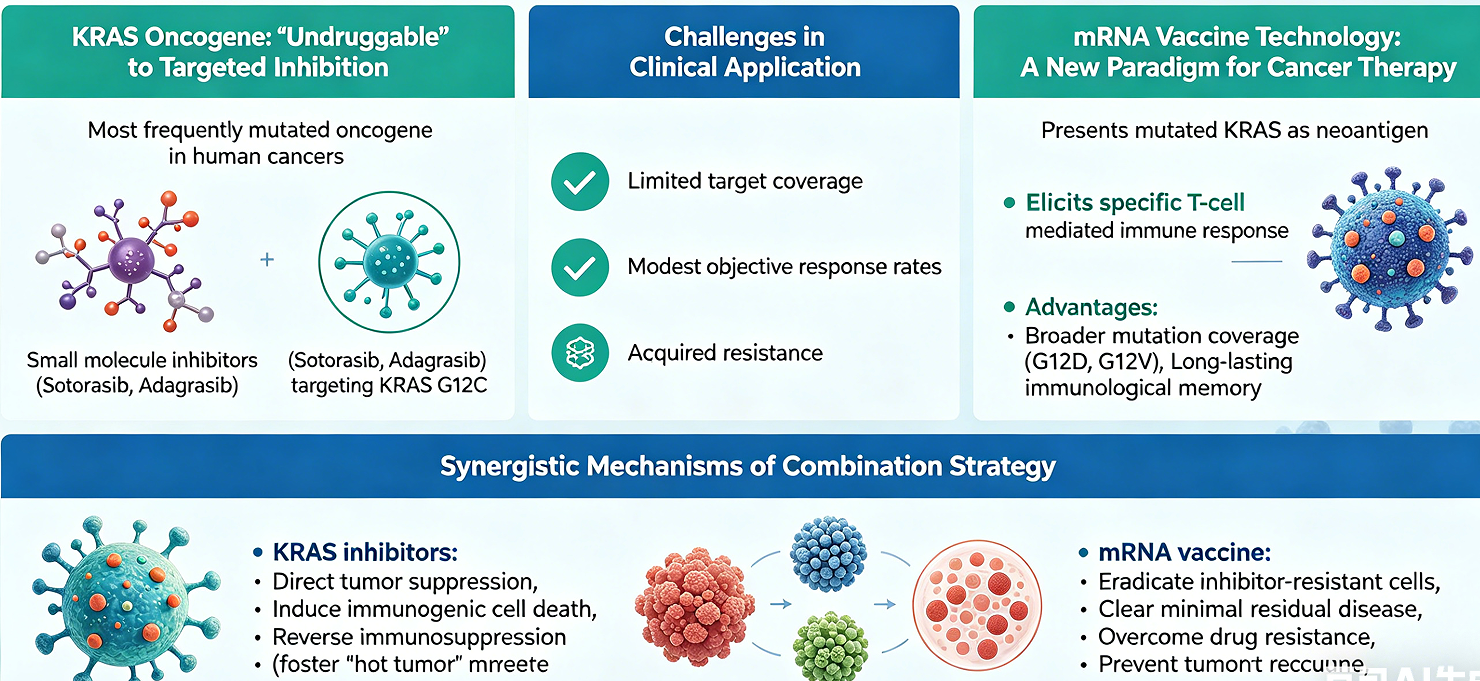
KRAS is one of the most frequently mutated oncogenes in human cancers and was long considered "undruggable." Although the successful development of small molecule inhibitors targeting the KRAS G12C mutation, such as Sotorasib and Adagrasib, represents a historic breakthrough in targeted therapy, their clinical application is hampered by significant challenges, including limited target coverage, modest objective response rates, and the inevitable development of acquired resistance. Concurrently, the success of mRNA vaccine technology in infectious disease prevention has validated its potential as a powerful platform, offering a new paradigm for cancer therapy. By presenting mutated KRAS as a neoantigen, mRNA cancer vaccines can elicit a specific T-cell mediated immune response to precisely eliminate tumor cells. This approach holds the unique advantages of covering a broader range of KRAS mutations (e.g., G12D, G12V) and inducing long-lasting immunological memory.
This article systematically elucidates an innovative "targeted therapy plus immunotherapy" combination strategy, integrating KRAS small molecule inhibitors with mutation-specific KRAS mRNA vaccines. The central thesis is that these two modalities can exert synergistic anti-tumor effects through complementary mechanisms. On one hand, KRAS inhibitors not only directly suppress tumor growth but may also foster a "hot tumor" microenvironment more permissive to T-cell infiltration by inducing immunogenic cell death and reversing immunosuppression. On the other hand, the mRNA vaccine can precisely eradicate tumor cells that have developed resistance to inhibitors via mechanisms such as bypass pathway activation, as well as clear minimal residual disease, thereby overcoming drug resistance and preventing tumor recurrence. By reviewing the mechanisms of action, clinical progress, and limitations of each therapy, this paper provides an in-depth analysis of the theoretical underpinnings for their synergistic combination, offering a strong rationale and a forward-looking perspective for the development of a more effective and durable treatment paradigm for KRAS-mutant cancers.
Downloads
Published
Issue
Section
License
Copyright (c) 2026 Advanced Interdisciplinary Science and Technology

This work is licensed under a Creative Commons Attribution 4.0 International License.




