Type 2 Diabetes Mellitus and Dietary Management: From Mechanisms to Personalized Strategies

Abstract
Type 2 diabetes is a rapidly increasing metabolic disease worldwide, driven by unhealthy dietary patterns, chronic inflammation, nutrient imbalances, sedentary behavior and disturbed circadian rhythm. This review summarizes mechanistic evidence linking high intakes of saturated fat, refined carbohydrates, and UPFs to insulin resistance through activation of inflammatory pathways, hepatic triglyceride accumulation, glucolipotoxicity, gut barrier impairment, and disturbances in gut microbiota. The roles of fiber, antioxidant nutrients, and micronutrients in preserving insulin sensitivity and β-cell function are highlighted. Major evidence-based dietary patterns, including the MD, DASH Diet, low-carbohydrate diets are also introduced. Other factors including intermittent fasting, lifestyle habits and psychological states are also mentioned in relation to blood sugar and lipid levels as well as metabolic risks. The review further discusses dietary strategies tailored to Chinese populations, established a dietary structure with cultural characteristics and emphasized traditional functional foods such as tea. Finally, we outline emerging applications of ML in diabetes care, including risk prediction, data identification and personalized dietary response. These insights support an integrated framework to improve long-term outcomes in T2D management.
Keywords: Type 2 diabetes; Dietary patterns; Insulin resistance; Precision nutrition; Machine learning
1 Introduction
Type Ⅱ diabetes (T2D) is one of the most prevalent metabolic disorders worldwide, characterized by persistent hyperglycemia and insulin resistance. According to the 11th Diabetes Atlas (IDF, 2024), 589 million people are now living with diabetes, and this number is projected to reach 853 million by 2050 [1]. Among the patients, over 75% live in low- and middle-income countries, where dietary imbalance and low nutrition literacy exacerbate the burden of disease.
Unhealthy dietary patterns, particularly high consumption of ultra-processed foods (UPFs), are the major drivers of global T2D epidemic. Each 10% increase in UPF intake causes the raises of diabetes risk by ≈15% [2]. In contrast, balanced dietary structures and regular meal timing stabilize glycemia, underscoring diet as the cornerstone of T2D prevention [3].
However, most current dietary recommendations rely on population averages while overlook the differences between individuals in aspects of metabolism, genetics, and cultural habits. Greater consumption of fast-food further widen the gap between guidelines and real-world diets at the same time. Therefore, developing region-specific, culturally adaptive, and personalized dietary strategies is imperative.
This review systematically analyzes the metabolic mechanisms and dietary patterns of T2D, evaluates major dietary models (e.g., Mediterranean, DASH, low-carbohydrate diets), and proposes innovative nutritional management strategies that are suitable for Chinese populations, adding ML-supported methods, aiming to promote precision nutrition and digital health approaches in diabetes prevention and care.
2 Etiology of T2D — Dietary Mechanisms and Metabolic Dysregulation
Insulin resistance and β-cell dysfunction are two core pathophysiological features of T2D and are closely associated with long-term dietary patterns and chronic overload of metabolism. Unbalanced diets disrupt energy metabolism, lipid signaling, inflammatory homeostasis, and gut microbiota composition, ultimately impairing glucose regulation and promoting metabolic dysregulation.
2.1 Effects of High-Fat and High-Sugar Diets
High-fat and high-sugar diets are usually major contributors to T2D epidemic, causing insulin resistance and metabolic dysfunction. Excessive intake of saturated fatty acids (SFA) activates inflammatory pathways of body mechanism, such as NF-κB and JNK. This increases macrophage infiltration in adipose tissue, while induces chronic low-grade inflammation at the same time. Thereby, insulin receptor phosphorylation and downstream signaling will be disrupted [4]. As shown in Figure 1, excessive saturated fatty acid intake induces inflammation and insulin resistance through macrophage activation and impaired insulin signaling in metabolic tissues.
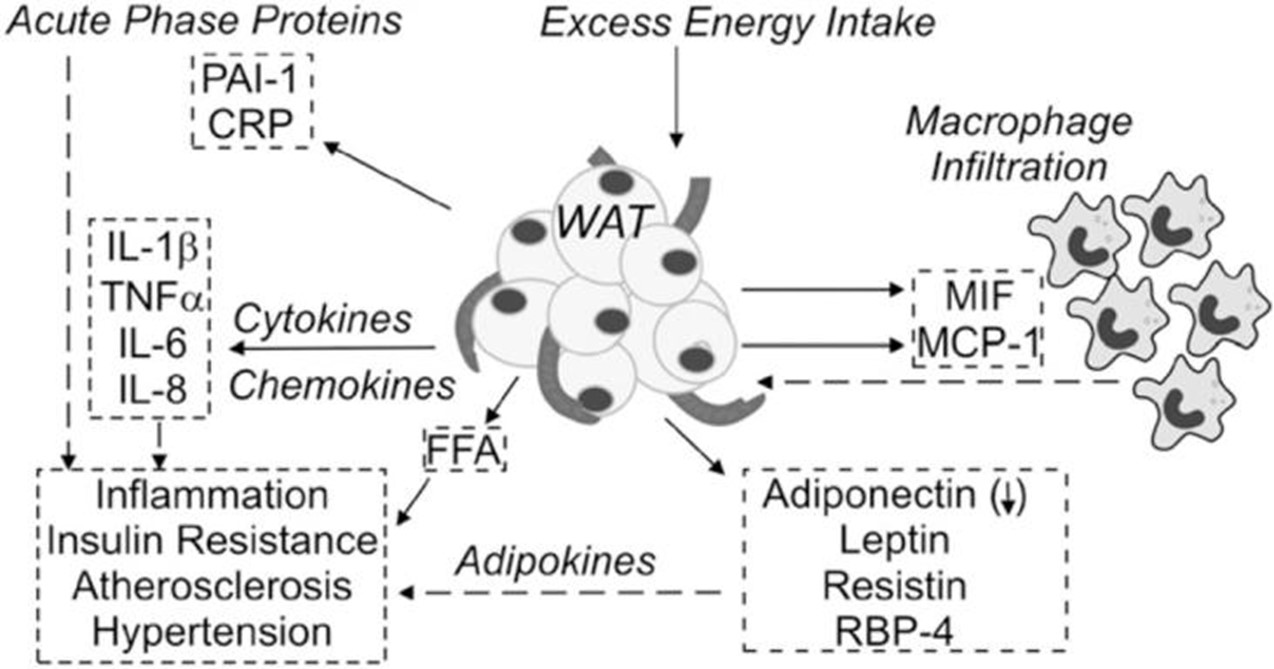
Hepatic triglyceride accumulation can be promoted by excessive fructose and refined carbohydrates consumption, this then cause non-alcoholic fatty liver disease (NAFLD) and visceral adiposity, further aggravate insulin resistance. Chronic exposure to high glucose and lipid levels also overburdens β-cells, leading to secretory dysfunction and apoptosis, accelerating disease progression [5].
The disadvantages of UPFs have been repeatedly noted in nutritional research. Prospective evidence suggests that a clear dose--response relationship: each additional serving of sugary beverages per day is associated with a roughly 27% increase in the risk of type 2 diabetes (RR 1.27, 95% CI 1.15--1.41) [7]. Collectively, these findings suggest that reducing the intake of saturated and fats and added sugars is a core strategy for preserving insulin sensitivity.
2.2 Imbalance of Other Macronutrients
Modern dietary patterns are featured by macronutrient imbalance, containing excessive refined carbohydrates such as white rice, wheat flour, and pastries. This foods pattern can cause rapid glucose spikes and maintained chronic hyperinsulinemia, which in turn suppress insulin receptor sensitivity and enhance lipogenesis. This actually forms a vicious cycle of "hyperinsulinemia--hyperglycemia--insulin resistance."
Different to carbohydrates, the consumption of protein has a bidirectional effect on glucose homeostasis. Short-term moderate high-protein diets can secrete PYY and GLP-1, thus enhance the satiety. This change further increases thermogenesis and improves control of glycemic. However, chronic overconsumption, particularly animal proteins, is related to elevated fasting glucose and insulin sensitivity reducing. However, great intake of plant-based and fish proteins can lower T2D incidence [8].
Another critical risk factor is insufficient dietary fiber consumption. Fibers moderates' carbohydrate absorption and nourishes gut microbiota. It also produces short-chain fatty acids (SCFAs) that improve insulin signaling and lower inflammation. On the other hand, low-fiber diets promote intestinal permeability and endotoxin translocation, causing chronic systemic inflammation [9]. Generally, high fat and sugar with low fiber diet remains a primary dietary driver of insulin resistance.
Antioxidant nutrients like Vitamin C and E can help neutralize free radicals, further reduce oxidative stress. It can protect β-cells and peripheral tissues against metabolic injury.
2.3 Micronutrient Deficiencies
Although T2D often exists combined with excessive energy intake, deficiencies in essential micronutrients also contribute significantly. Magnesium, chromium, and zinc are necessary cofactors in insulin synthesis and receptor activation. The deficiency of them also impairs glucose uptake and worsens insulin resistance. For example, Vitamin D enhances insulin receptor expression and PPAR-γ activation, improving β-cell function and glucose condition [10].
2.4 Lifestyle and Behavioral Factors
Lack of physical exercise or long-time sitting is another reason of lower insulin signaling. Such sedentary behavior increases the risk of T2D by over 20% for each additional hour of sitting. Sleep deprivation and circadian disruption are common in modern fast-paced living as well, altering insulin, melatonin rhythms, and lead to glucose dysregulation. In addition to lifestyle habits, psychological stress elevates cortisol levels and promote central adiposity very significantly. Moreover, irregular eating habits such as late-night eating also disturb gut microbiota and cause same result. In sum, an integrated lifestyle strategy combining regular movement, sufficient sleep, circadian alignment, and stress reduction is fundamental for T2D prevention and management.
3. Dietary Management Patterns in T2D
3.1 International Mainstream Dietary Patterns
Several evidence-based dietary patterns have already been established for T2D, offering benefits in many aspects. Mediterranean Diet (MD), DASH Diet, low-carbohydrate diet (LCD), and intermittent fasting (IF) are the most studied ones. A comparison of major dietary patterns and their metabolic effects in T2D is summarized in Table 1.
| Dietary Pattern | Core Characteristics | Key Outcomes | Limitations | References |
|---|---|---|---|---|
| MD | High MUFA (olive oil, nuts, fish, fruits, legumes, whole grains); low red meat & sugar | ↓ HbA1c ~0.39%, ↓ waist 1.7 cm, ↓ SBP ~4 mmHg; ↑ insulin sensitivity | Requires access to olive oil & fish; cost | [11]-[12] |
| DASH Diet | High fruits, vegetables, whole grains, low-fat dairy; low sodium & SSB | ↓ FBG ~11 mg/dL, ↓ BP, ↑ lipid & insulin profiles | Moderate adherence; high prep effort | [13]-[14] |
| LCD | <45% energy from carbs; ↑ protein & healthy fat | ↓ HbA1c ~0.28%, ↑ HDL, ↓ TG | Difficult adherence; risk of fiber deficiency | [15] |
| IF | Periodic fasting (e.g., 16:8, 5:2) or time-restricted feeding | ↓ HbA1c ~7 mmol/mol, ↓ weight ~4.8 kg, ↑ insulin sensitivity | Long-term safety unclear; adherence variable | [16] |
3.2 Dietary Patterns Suitable for the Chinese Population
Chinese diets are predominantly cereal-based, with refined grains still dominant, affecting glycemic control. Evidence shows that optimizing staple food structure with increased whole grains, legumes, and resistant starch can reduce T2D risk by ~37% when replacing 100 g/day of refined grains [17]. Traditional plant-based proteins and tea polyphenols also improve insulin sensitivity by modulating glucose absorption and gut microbiota [18].
The change of dietary pattern should respect regional diet cultures, like wheat-based diets in northern China and rice-based in southern regions. Meanwhile feasible substitutions like mixed-grain rice or resistant starch should be added as promoting. Overall, focusing on staple optimization of macro-nutrients and functional traditional foods can improve long-term metabolism in Chinese population.
3.3 Individualized and Precision Nutrition Management
With the development in metabolomics, microbiomics and digital health, dietary guidelines for individuals and populations are evolved in personalized nutrition [19]. In T2D management, individualized plans should consider the condition of BMI, insulin resistance, gut microbiota and circadian rhythm. For instance, obese individuals with high insulin resistance benefit from low-carbohydrate diets, whereas those with postprandial hyperglycemia respond to low-GI and rich SCFA producing diets.
Data platforms integrating clinical indices, CGM, dietary logs, and multi-omics enable ML-based prediction of PPGR for tailored interventions [20]-[21]. Precision nutrition thus represents both a research frontier and a clinical application pathway. However, this success depends on data privacy, model interpretability, and long-term validation in diverse populations.
4 Application of Machine Learning in Diabetes Dietary Prediction and Management
ML algorithms like Logistic Regression, Random Forest, XGBoost, and Neural Networks nowadays show remarkable potential for predicting T2D risk by modeling complex nonlinear interactions among dietary, metabolic, and genetic variables [22]. Tree-based models like Random Forest and XGBoost consistently outperform others; using the Pima Indians dataset, XGBoost achieved an accuracy of 77.83% and an AUC of 0.822. SHAP-based interpretability identified glucose concentration, BMI, and age as primary risk factors, demonstrating how explainable ML enhances individualized dietary assessment and clinical decision-making [23].
Unsupervised algorithms, including K-means clustering, principal component analysis (PCA), and reduced-rank regression (RRR), have been widely applied to identify dietary patterns linked to T2D risk. For example, Beigrezaei et al. reported that diets high in fruits and vegetables but low in processed meat and refined grains reduced T2D risk by approximately 46% [24]. Interpretable decision-tree models further visualize high-risk dietary combinations---such as low fruit and vegetable intake with high refined carbohydrates---providing quantitative evidence for dietary interventions and public health strategies.
In recent years, AI-assisted nutrition systems have evolved toward precision health management. Integrating ML with real-time dietary logs, continuous glucose monitoring (CGM), and wearable sensors enables dynamic prediction of postprandial glucose responses (PPGR), supporting personalized dietary feedback and closed-loop control [25]. These data-driven systems are reshaping digital nutrition management and represent a key direction for future diabetes care and prevention.
5 Innovative Dietary Recommendations
Effective dietary interventions for T2D should adhere to universal nutritional principles while adapting to traditional Chinese dietary structures, combining ML-based predictive tools and public health statistic at the same time.
Guidelines should emphasize energy balance, low-GI, higher fiber, plant-based or lean fish protein and replacement of saturated with polyunsaturated fats, while limiting sugar beverages and UPFs.
For Chinese, improving staple quality through whole grains and RS can reduce glycemic load and enhance insulin sensitivity, aligning with the Chinese Dietary Guidelines [26]. Integrating CGM and wearable sensor technology into ML-driven platforms enables more reliable prediction of glucose responses and individual dietary feedback.
6 Conclusion and Future Perspectives
T2D is a major metabolic disease, strongly influenced by diet pattern, energy balance, and lifestyle. Optimizing the quality of carbohydrate, increasing dietary fiber and polyunsaturated fats, and ensuring balance micronutrients can enhance insulin sensitivity, decrease inflammation, and stabilize glycemia.
Evidence-based dietary patterns, such as the MD, DASH, and low-carbohydrate diets, can consistently improve metabolic markers, even though the effects may vary depending on regional food culture and individual metabolic status. For the Chinese population, strengthening a plant-based, high-fiber diet and reducing refined sugar and saturated fat intake can improve long-term metabolic health.
The convergence of machine learning and digital health will drive the development of a new generation of precision nutrition, enabling dynamic prediction of blood sugar response and personalized dietary recommendations. Future research should focus on multicenter trials, omics integration, and the translation of clinical applications of AI-based nutrition platforms to transform diabetes care from experience-driven to data-driven precision management.


