A Theranostic Nanorobotic Platform for Cancer: Integrating Smart Nanomaterials and Biological Computers into Tumor Microenvironment-Responsive Systems
Abstract
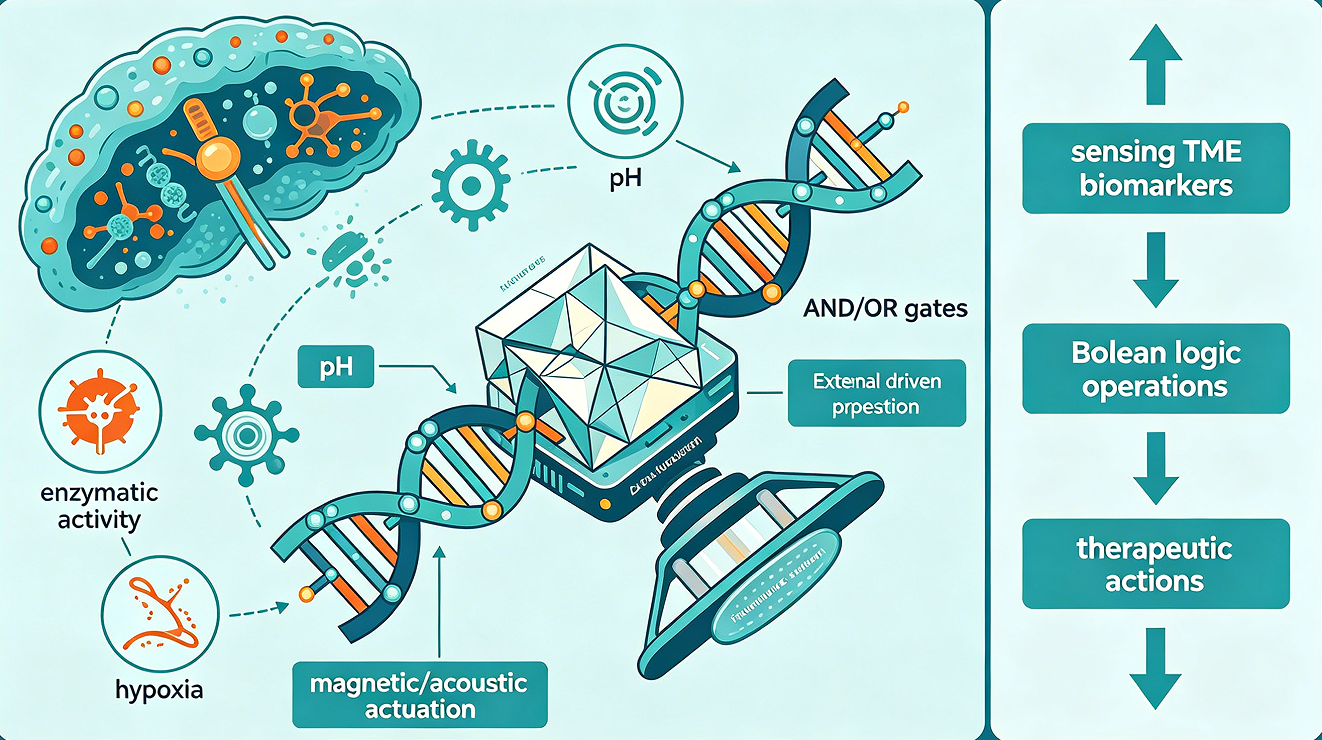
Conventional nanomedicine approaches for cancer therapy face a fundamental limitation: they lack the capacity for autonomous sensing and decision-making, rendering them incapable of adapting to the dynamic and heterogeneous nature of the tumor microenvironment (TME). This review presents a comprehensive framework for a next-generation theranostic nanorobot that integrates three transformative technologies: DNA origami for structural precision, nucleic acid-based logic gates for onboard computing, and externally actuated propulsion systems for targeted navigation. We propose a closed-loop architecture wherein the nanorobot senses multiple TME biomarkers — including pH, enzymatic activity, and hypoxia — processes these inputs through Boolean logic operations (AND/OR gates), and executes therapeutic actions exclusively upon satisfaction of predefined criteria. This paradigm shifts from passive accumulation to active computation, enabling unprecedented specificity in tumor targeting. We critically examine recent advances in DNA nanotechnology for logic-gated molecular systems, magnetic and acoustic actuation platforms for in vivo navigation, and optoacoustic imaging modalities for real-time tracking. Key challenges addressed include biocompatibility, bioenergetics for autonomous function, and integration of synthetic biology tools for metabolic support. Finally, we envision a future "sense-compute-respond" platform that embodies the convergence of nanobiotechnology, systems biology, and precision medicine.
Keywords: Nanorobotics · DNA origami · Logic gates · Tumor microenvironment · Theranostics · Optoacoustic imaging · Synthetic biology
1. Introduction
The advent of nanomedicine has revolutionized cancer therapeutics, yet a persistent chasm remains between the theoretical promise of targeted drug delivery and its clinical realization. Nanoparticulate systems — liposomes, polymeric nanoparticles, and inorganic carriers — have demonstrated improved pharmacokinetics and passive tumor accumulation via the enhanced permeability and retention (EPR) effect. However, these first-generation nanocarriers are fundamentally passive entities: they lack the capacity to perceive their biochemical surroundings, process multiple streams of information, or make autonomous decisions. This limitation becomes critically consequential in the context of the tumor microenvironment (TME), a dynamic and heterogeneous ecosystem characterized by regional variations in pH, oxygen tension, enzymatic composition, and metabolic flux.
The TME presents not a static target but an evolving landscape. Solid tumors exhibit pronounced spatial and temporal heterogeneity: hypoxia gradients shift with vascular density, extracellular pH varies from mildly acidic (pH 6.5–6.9) in peripheral regions to profoundly acidic (pH < 6.0) in necrotic cores, and matrix metalloproteinase (MMP) concentrations correlate with invasive fronts. A nanocarrier that releases its payload solely in response to low pH may activate prematurely in inflamed but non-malignant tissues; one that recognizes a single surface marker may target healthy cells expressing that marker at baseline levels. The clinical consequences of such imprecision are not merely academic — they manifest as dose-limiting toxicities, suboptimal therapeutic indices, and the propagation of resistant clonal populations.
What is required, therefore, is a fundamental reimagining of the therapeutic nanoparticle: not as a passive cargo vessel, but as an autonomous agent capable of sensing, computing, and responding. This vision aligns with the emerging paradigm of "theranostic nanorobotics" — integrated systems that combine diagnostic sensing with therapeutic execution within a single programmable platform. The enabling technologies for such a system are now coalescing at the intersection of DNA nanotechnology, synthetic biology, and advanced imaging.
DNA origami, first demonstrated by Rothemund in 2006, provides a molecular-scale scaffolding platform with unprecedented programmability. Through the precise folding of a long scaffold strand by hundreds of short staple strands, DNA origami enables the construction of nanostructures with arbitrary geometries — nanosprings, cages, rods, and more complex architectures — with feature sizes approaching 2 nm. The sequence-specific addressability of DNA allows for the spatial organization of functional moieties with molecular precision, including therapeutic payloads, targeting ligands, and — crucially for our purposes — computational elements.
The computational elements themselves derive from the field of DNA computing, which exploits the predictable base-pairing thermodynamics of nucleic acids to implement Boolean logic operations. Toehold-mediated strand displacement, catalytic hairpin assembly (CHA), and hybridization chain reaction (HCR) have been harnessed to construct AND, OR, NOT, and more complex logic gates that process molecular inputs. When integrated with aptamer-based sensors for TME biomarkers, these molecular circuits enable the nanorobot to perform "if-and-only-if" computations: only when input A (e.g., low pH) AND input B (e.g., MMP-9) are simultaneously present does the system output a therapeutic response.
The third enabling technology is actuation. While Brownian motion dominates at the nanoscale, directed movement requires exogenous energy input. Magnetic fields offer a particularly attractive actuation modality: they penetrate deeply into tissue, are non-invasive, and pose minimal safety concerns at clinically relevant strengths. Recent advances in magnetic microrobot design have demonstrated upstream swimming against flow rates exceeding 70 mm/s, swarm reconfiguration for navigating vascular bifurcations, and targeted accumulation in specific vessel branches. Complementary acoustic propulsion systems, leveraging oscillating microbubbles, have achieved real-time ultrasound tracking with sub-millimeter precision.
This review synthesizes these disparate threads into a unified framework: a TME-responsive theranostic nanorobot that embodies "sense-compute-respond" autonomy. We proceed as follows. Section 2 establishes the design principles and architectural requirements. Section 3 details the implementation of sensing and logic-gated computation using DNA nanotechnology. Section 4 addresses actuation strategies for targeted navigation and real-time imaging. Section 5 confronts the challenges of biocompatibility, bioenergetics, and in vivo validation. Section 6 concludes with an outlook toward clinical translation and future directions.
2. System Architecture and Design Principles
2.1 The "Sense-Compute-Respond" Paradigm
The proposed nanorobot operates according to a closed-loop control architecture comprising three functional modules. The sensing module comprises molecular recognition elements — typically aptamers or DNAzyme-based sensors — that bind specifically to TME biomarkers with measurable affinity. The computational module processes the binary or graded signals from multiple sensors through logic gates, generating a decision output only when the input pattern matches a predefined therapeutic threshold. The response module transduces this decision into a therapeutic action, which may include drug release, photothermal conversion, or the generation of cytotoxic species.
This architecture confers three critical advantages over conventional nanocarriers. First, specificity enhancement: by requiring multiple independent biomarkers for activation, the system dramatically reduces off-target activation. A single biomarker may be elevated in both cancerous and inflammatory conditions; the conjunction of low pH, elevated MMP, and hypoxia is far more pathognomonic for malignancy. Second, adaptability: the computational module can be reprogrammed by altering the DNA sequences that implement the logic functions, allowing rapid iteration as biomarker profiles are refined. Third, autonomy: the nanorobot makes decisions locally, without requiring external instruction, which is essential for treating disseminated micrometastases or regions beyond the reach of real-time imaging guidance.
2.2 Biomarker Selection and Logic Specification
The choice of input biomarkers critically determines the specificity and sensitivity of the system. An optimal biomarker set should satisfy several criteria: (1) differential expression between tumor and normal tissues; (2) accessibility from the vascular compartment or extracellular space; (3) measurability by nucleic acid-based sensors; and (4) orthogonal regulation to minimize crosstalk.
Based on extensive characterization of the TME, we propose a three-input panel: extracellular pH, matrix metalloproteinase 2 (MMP-2) activity, and hypoxia-inducible factor 1-alpha (HIF-1α) concentration. Each of these parameters exhibits characteristic alterations in solid tumors and has been successfully targeted by DNA-based sensors.
pH is a particularly attractive input because of its broad dynamic range and spatial heterogeneity. The extracellular pH of normal tissues is tightly maintained at 7.35–7.45; solid tumors, due to the Warburg effect and inadequate perfusion, exhibit extracellular acidification to pH 6.0–6.8. DNA-based pH sensing exploits the i-motif structure — a non-canonical quadruplex formed by cytosine-rich sequences under acidic conditions. The i-motif folds at pH < 6.5 and dissociates at neutral pH, providing a reversible, cooperative switching mechanism with a sharp transition near pH 6.0–6.5. Split i-motif designs, in which two hemiduplexes assemble only upon protonation, enable AND-gated logic when combined with a second input.
MMP-2, a zinc-dependent endopeptidase overexpressed in invasive tumor fronts, degrades basement membrane collagen IV and facilitates metastasis. Peptide substrates cleaved specifically by MMP-2 have been incorporated into DNA-peptide conjugates; proteolytic cleavage releases a DNA fragment that triggers strand displacement cascades. This design ensures that the enzymatic activity, rather than mere presence, is detected — a critical feature given that MMP-2 is secreted as an inactive zymogen requiring activation.
HIF-1α, the master transcriptional regulator of the hypoxic response, is stabilized under low oxygen tension (pO₂ < 5 mmHg) and degraded under normoxia. While primarily intracellular, secreted exosomal HIF-1α and HIF-regulated proteins (e.g., carbonic anhydrase IX) provide accessible surrogates. Aptamers targeting CA-IX have been developed with nanomolar affinity and incorporated into logic-gated constructs.
The Boolean logic function must be specified based on the desired specificity-sensitivity trade-off. An AND gate (Input A AND Input B AND Input C) maximizes specificity but may reduce sensitivity if any input falls below threshold in some tumor regions. An OR gate (Input A OR Input B OR Input C) maximizes sensitivity at the cost of specificity. A hybrid architecture — OR-gated sensing of three biomarkers feeding into an AND-gated therapeutic release — offers a balanced approach, as recently demonstrated by Ling et al. using Pd@Pt nanoparticle-DNA nanomachines. In their design, the detection of any of three miRNA biomarkers triggers a fluorescent signal (OR logic), but the photothermal therapeutic output requires all three inputs (AND logic), implementing a "diagnostic OR, therapeutic AND" strategy.
2.3 Architectural Integration
The physical integration of sensing, computing, and response modules within a single nanoscale construct presents formidable challenges. The DNA origami platform offers a compelling solution due to its structural programmability and spatial addressability. A typical DNA origami nanorobot measures 50–100 nm in diameter, a size regime that permits extravasation through fenestrated tumor vasculature while avoiding rapid renal clearance.
Karna et al. recently demonstrated a DNA origami nanospring system incorporating Boolean logic gates responsive to multiple miRNA inputs. The nanospring — a 3D coiled structure approximating 400 nm in contour length — underwent mechanical uncaging upon logic satisfaction, exposing RGD ligands that engaged integrin receptors on cancer cell surfaces. This work established that DNA nanostructures can transduce molecular computations into mechanical outputs, a principle extensible to drug release.
A hierarchical integration strategy is proposed: the DNA origami scaffold serves as the chassis, with sensor aptamers displayed on the exterior surface for optimal biomarker access. The logic computation occurs through strand displacement reactions localized within the interior cavity, protected from nuclease degradation by a permeable but sterically hindering shell. The therapeutic payload — chemotherapeutic small molecules, photosensitizers, or immunomodulators — is loaded into the cavity or conjugated via cleavable linkers controlled by the logic output. Magnetic nanoparticles (e.g., FePt or Fe₃O₄) are embedded within the scaffold for actuation and imaging contrast.
3. DNA-Based Sensing and Logic Computation
3.1 Molecular Recognition Elements for TME Biomarkers
The fidelity of the sensing module determines the overall specificity of the nanorobot. Among the various molecular recognition strategies available — antibodies, peptides, small molecules, and nucleic acids — aptamers offer unique advantages for DNA-based computing systems. Aptamers are single-stranded DNA or RNA oligonucleotides selected by systematic evolution of ligands by exponential enrichment (SELEX) to bind target molecules with high affinity and specificity. Their sequence-encoded nature allows direct integration into DNA circuits without chemical conjugation.
For pH sensing, the i-motif remains the most versatile platform. I-motifs are formed by C-rich sequences that fold into a four-stranded structure stabilized by hemiprotonated C–C base pairs under acidic conditions. The folding transition exhibits strong cooperativity and tunable pH thresholds through sequence variation. Ling et al. employed a split i-motif design in which two separate DNA strands each contain half of the i-motif sequence; only upon protonation do the strands assemble into a complete i-motif, bringing their appended functional domains into proximity. This split design ensures that pH sensing is coupled to — rather than independent of — other inputs.
For protein biomarkers, aptamers selected against specific targets provide the recognition interface. The Sgc8c aptamer, which binds protein tyrosine kinase 7 (PTK7) overexpressed on multiple cancer cell lines (e.g., CCRF-CEM leukemia cells), has been extensively characterized with a dissociation constant (K_d) of approximately 0.8 nM. For MMP-2, peptide substrates offer superior specificity compared to aptamers, as they report directly on enzymatic activity. The peptide sequence PLGLAG (cleaved between G and L) is preferentially cleaved by MMP-2 over other MMP family members; conjugation to DNA via maleimide-thiol chemistry enables integration with strand displacement circuits.
3.2 Logic Gate Implementation via Toehold-Mediated Strand Displacement
Toehold-mediated strand displacement is the fundamental reaction underlying most DNA logic circuits. A toehold is a short (5–8 nucleotide) single-stranded region on a double-stranded DNA complex that serves as an initiation site for an invading strand. The invading strand hybridizes to the toehold, then undergoes branch migration — a process of sequential base exchange — to displace the incumbent strand. The reaction rate is tunable over five orders of magnitude by varying toehold length and sequence, enabling complex kinetic control.
Catalytic hairpin assembly (CHA) amplifies the output of strand displacement reactions. In CHA, two metastable hairpin structures (H1 and H2) coexist without interacting in the absence of a catalyst. An input strand (the catalyst) opens H1 by binding to its toehold; the exposed region of H1 then opens H2, forming a stable H1–H2 duplex and releasing the input strand to catalyze additional reactions. This amplification mechanism converts a single molecular recognition event into a large structural change, enhancing sensitivity.
Ling et al. demonstrated an AND-gated CHA system for cancer cell detection. Their design required two inputs: PTK7 (detected by the Sgc8c aptamer) and low pH (detected by split i-motif assembly). Only when both conditions were satisfied did the CHA trigger fragment become exposed, initiating amplification on an electrode surface and generating an electrochemical signal detectable at a limit of 15 cells/mL. This work validates the feasibility of AND-gated activation in complex biological matrices (fetal bovine serum and cell culture media), an essential step toward in vivo translation.
For the proposed nanorobot, a three-input AND gate can be implemented by cascading two-input gates. One approach is to use a "proximal" design in which three sensor modules are tethered to a common DNA nanostructure; all three must be activated to assemble a complete trigger strand that initiates the therapeutic response. Alternatively, a "concatenated" design uses the output of one gate as the input to the next, analogous to electronic logic circuits. The latter approach offers greater design modularity but may suffer from signal attenuation through successive gates.
3.3 Recent Advances: From Single Gates to Integrated Circuits
The field has progressed rapidly from simple two-input gates to multi-input, reconfigurable circuits. Karna et al. demonstrated DNA origami nanosprings that implement both AND and OR gates responsive to miRNA inputs. Mechanical uncaging — the exposure of RGD ligands previously sterically blocked by the nanospring's coiled conformation — occurred only upon logic satisfaction. This mechanical transduction mechanism is particularly relevant for drug delivery, as it enables the presentation of targeting ligands in a conditional manner: the nanorobot remains "stealthy" during circulation but becomes adhesive upon reaching the TME.
A parallel effort by a separate research group described a continuous-input DNA nanomachine that programs automated tumor diagnosis and treatment. Their Pd@Pt octapod nanoparticle-DNA conjugate integrated OR and AND logic gates with three miRNA biomarkers (miR-21, miR-155, and miR-196a). The OR gate provided early warning (fluorescent signal upon any biomarker detection), while the AND gate (requiring all three) triggered 808 nm laser-induced photothermal therapy. The Pd@Pt nanoparticles also exhibited catalase-like nanozyme activity, converting endogenous H₂O₂ to O₂ to alleviate tumor hypoxia — an elegant example of therapeutic augmentation beyond direct cytotoxicity.
These advances collectively demonstrate the feasibility of DNA-based molecular computation within nanoscale platforms. The remaining challenges — discussed in Section 5 — pertain to nuclease stability, in vivo signal-to-noise ratio, and the scalability of DNA synthesis for clinical-grade production.
4. Actuation and Imaging: Navigation and Real-Time Tracking
4.1 Magnetic Actuation for Targeted Navigation
For a nanorobot to reach its intended target, passive diffusion is insufficient. The vasculature presents a dynamic environment: blood flow velocities range from 1–10 mm/s in capillaries to 100–400 mm/s in arteries; shear rates vary by orders of magnitude; and vascular geometries include bifurcations, aneurysms, and stenoses. Directed navigation against such flow requires active propulsion.
Magnetic actuation has emerged as the leading strategy for in vivo microrobot control due to its deep tissue penetration, rapid response, and established safety profile. A recent breakthrough by Nozdriukhin et al. demonstrated magnetic microflowers — copper phosphate particles decorated with iron oxide nanoparticles and indocyanine green — capable of precise optoacoustic localization and intravascular magnetic actuation. Their system achieved real-time tracking of individual 5–20 μm particles in mouse brain vasculature using super-resolution optoacoustic imaging, with magnetic fields steering particles into specific vascular branches.
At the nanoscale, the FePt-ZIF-8 core-shell system reported by Nanowerk exemplifies the integration of magnetic actuation with drug delivery. The FePt core provides high saturation magnetization (24.70 emu/g) and coercivity (153.68 mT), enabling stable steering even after removal of the external field. The ZIF-8 shell, a metal-organic framework, serves both as a drug reservoir (93.9% doxorubicin loading efficiency) and a pH-responsive release mechanism: at pH 5.5, approximating the TME, near-complete drug release occurs. Importantly, the particles formed reconfigurable swarms — chains, vortices, or diffuse clouds — depending on the applied field pattern, allowing upstream movement against flow rates of 70 mm/s and selective navigation into branched vessels with 90.5% accuracy.
The swarm behavior observed in magnetic nanoparticle ensembles is particularly significant for clinical translation. Individual nanoparticles experience Brownian forces that may randomize their trajectories; however, when assembled into swarms via oscillating or rotating fields, the collective magnetic torque overcomes thermal fluctuations, enabling deterministic control. Moreover, swarms can be dispersed by altering the field pattern, preventing vascular occlusion — a critical safety feature.
4.2 Acoustic Propulsion and Ultrasound Imaging
Magnetic actuation, while powerful, requires the generation of field gradients that diminish with distance from the electromagnet. Acoustic propulsion offers a complementary modality: ultrasound waves penetrate deeply into tissue, are non-ionizing, and can be focused with high spatial resolution.
Han et al. developed bioresorbable acoustic microrobots (BAMs) fabricated via two-photon polymerization of PEGDA-PETA hydrogels. The key innovation is a dual-opening bubble-trapping cavity: the hydrophobic inner surface retains microbubbles for multiday durations in biological fluids, while the hydrophilic outer surface prevents aggregation. Under focused ultrasound stimulation (frequencies in the MHz range), the entrapped microbubbles oscillate, generating microstreaming vortices that propel the microrobot. Speeds exceeding 2 mm/s were achieved in PBS, with stable propulsion in urine, gastrointestinal fluid, and whole blood.
The same microbubbles that enable propulsion also serve as ultrasound contrast agents. The BAMs were visualized in real time using clinical ultrasound probes in a murine bladder tumor model, with magnetic navigation (Fe₃O₄ nanoparticles embedded in the hydrogel) directing the microrobots to the tumor site. Following therapy, the hydrogel degraded via hydrolysis, eliminating the risk of long-term retention. This combination of acoustic propulsion, ultrasound imaging, and biodegradability represents a significant step toward clinical translation.
4.3 Optoacoustic Imaging for Deep Tissue Tracking
While ultrasound provides excellent spatial resolution and real-time feedback, its contrast mechanism (acoustic impedance) may not distinguish nanorobots from surrounding tissue with sufficient specificity. Optoacoustic (photoacoustic) imaging addresses this limitation by combining the contrast of optical absorption with the penetration depth of ultrasound.
Aziz et al. systematically characterized nanomaterial-decorated micromotors for enhanced photoacoustic imaging. Gold nanorods and nanostars, which exhibit strong localized surface plasmon resonance in the near-infrared (NIR) window (700–900 nm), were conjugated to magnetic micromotors. The NIR window is optimal for deep tissue imaging because hemoglobin and water absorption minima coincide, enabling imaging depths of several centimeters. Through COMSOL simulations and experimental validation, the authors demonstrated that increasing the spacing between gold nanostructures and the underlying oxide layer enhanced light absorption and preserved the characteristic absorption peak — critical for distinguishing microrobots from endogenous chromophores.
The FePt-ZIF-8 system described previously also functions as an optoacoustic contrast agent. The FePt core absorbs strongly at 920 nm, a wavelength where blood absorption is minimal. Using multispectral optoacoustic tomography (MSOT), the particles remained visible through 7 cm of porcine brain tissue. When combined with localization optoacoustic tomography — a super-resolution technique that localizes individual particles by fitting pixel intensities to effective point spread functions — the system resolved vascular structures as narrow as 22 μm at depths of 1.5 mm through intact scalp and skull.
The convergence of magnetic actuation and optoacoustic imaging in a single particle platform is particularly powerful. Magnetic fields provide coarse navigation (directing the swarm to the tumor-bearing region), while optoacoustic imaging enables fine tracking of individual particles. Closed-loop control — adjusting the magnetic field based on real-time imaging feedback — is theoretically achievable but has not yet been demonstrated in vivo.
4.4 Towards Closed-Loop Guidance
The ultimate actuation-imaging system would incorporate closed-loop feedback: the imaging modality continuously reports nanorobot position; a control algorithm computes the required field adjustments; and the actuation system executes these adjustments. This capability has been demonstrated in vitro but remains challenging in vivo due to the latency between image acquisition, processing, and field adjustment, as well as the need to integrate multiple imaging modalities (optoacoustic for deep tracking, ultrasound for real-time feedback, and MRI for anatomical reference).
5. Technical Challenges and Emerging Solutions
5.1 Biocompatibility and Immunogenicity
For any nanorobot intended for clinical use, biocompatibility is paramount. The constituent materials — DNA, magnetic nanoparticles, metal-organic frameworks, and hydrogels — must be either biodegradable or cleared from the body without toxicity.
DNA is intrinsically biocompatible, as it is composed of endogenous nucleotides. However, unmodified DNA is rapidly degraded by serum nucleases, with a half-life of minutes to hours in vivo. Several stabilization strategies exist: chemical modifications (phosphorothioate backbones, 2'-O-methyl ribose) enhance nuclease resistance while preserving base-pairing fidelity; encapsulation within protective shells (e.g., lipid bilayers or polymer coatings) shields the DNA from enzymatic attack; and the dense packing of DNA within origami structures confers some steric protection.
The magnetic core materials require more careful consideration. Iron oxide (Fe₃O₄) nanoparticles have an established safety profile: they are metabolized by endogenous iron pathways and cleared with half-lives of days to weeks. FePt, while offering superior magnetic properties, raises concerns about platinum toxicity. The study by Nanowerk reported that FePt-ZIF-8 particles (200 nm diameter) showed minimal cytotoxicity (87% viability in neuronal cells at 10 μg/mL) and only transient inflammatory responses. However, long-term biodistribution studies are needed to rule out accumulation in the reticuloendothelial system.
The hydrogel BAMs described by Han et al. address biocompatibility through design: the PEGDA-PETA matrix degrades via hydrolysis over 2–4 weeks, releasing non-toxic degradation products. The degradation rate is tunable by adjusting the crosslinking density and incorporating hydrolytically labile segments.
5.2 Bioenergetics: Powering Autonomous Function
A critical but often overlooked challenge is energy: how does a nanoscale device perform computation and actuation without an onboard power source? DNA strand displacement is thermodynamically driven by the free energy of base-pair formation; no external energy input is required for computation per se. However, the system is not at equilibrium — it requires a source of "fuel" to drive reactions forward and prevent backflow.
In DNA circuits, fuel is typically provided by "waste" duplex formation: the displacement reaction releases a single strand that may be less stable than the product duplex. The free energy difference (ΔG) between initial and final states drives the reaction forward. For a typical 8-nucleotide toehold, ΔG is approximately -15 to -20 kcal/mol, sufficient to drive the reaction under physiological conditions. However, as the circuit performs multiple operations, the system approaches equilibrium and eventually stalls. Continuous-input designs, such as the one described by a research group, address this by supplying fresh input strands over time.
For actuation, magnetic and acoustic fields provide exogenous energy. This is advantageous from a safety perspective — the nanorobot is inert in the absence of the field — but limits autonomous operation to field-accessible regions. A hybrid approach, in which the nanorobot harvests energy from endogenous sources (e.g., glucose or ATP), would represent a significant advance.
Kim et al. recently reported an autonomous chemo-metabolic system for constructing cell-in-shell nanobiohybrids that may offer a path forward. Their system harnessed ethanol fermentation by Saccharomyces cerevisiae, chemically coupled with an enzymatic cascade (alcohol oxidase and horseradish peroxidase), to drive polydopamine nanoshell formation. While demonstrated in yeast rather than mammalian systems, the principle — coupling cellular metabolism to nanorobot function — could be adapted. For example, a nanorobot equipped with glucose oxidase and catalase could generate oxygen from endogenous glucose, alleviating hypoxia while powering actuation via oxygen bubble generation.
5.3 In Vivo Validation and Preclinical Models
Translating from benchtop demonstrations to in vivo function requires rigorous validation in progressively complex models. The current state of the art includes:
In vitro: microfluidic channels mimicking vascular geometries, with controlled flow rates and biomarker gradients. The magnetic swarm navigation studies achieved 90.5% targeting accuracy in branched channels.
Ex vivo: tissue slices or isolated organs. Optoacoustic imaging through 7 cm of porcine brain tissue demonstrated the penetration depth achievable with NIR-absorbing particles.
In vivo rodent models: subcutaneous or orthotopic tumors. BAMs were tested in a murine bladder tumor model, with ultrasound-guided magnetic navigation and subsequent tumor size reduction. Magnetic microflowers were tracked in mouse brain vasculature using optoacoustic imaging.
Critical gaps remain. No study to date has demonstrated a fully integrated "sense-compute-respond" nanorobot in a living animal — one that senses TME biomarkers, performs logic computation, and releases drug only upon gate satisfaction. Most studies focus on individual subsystems: logic-gated sensing in vitro, magnetic actuation in vivo, or imaging in ex vivo tissues. The integration of all three in a single animal study represents the next major milestone.
5.4 Scalability and Manufacturing
The synthesis of DNA origami at scale remains challenging. A typical origami preparation uses 10–20 μg of scaffold strand (M13mp18 bacteriophage genome) per 100 μL reaction, sufficient for laboratory studies but orders of magnitude below clinical needs. Rolling circle amplification (RCA) and enzymatic production methods are being developed to produce scaffold strands at lower cost. Staple strands, synthesized by solid-phase oligonucleotide synthesis, are the primary cost driver: a typical origami requires 200–300 unique staples at the 100 nmol scale, costing thousands of dollars per milligram of origami. Advances in microarray-based synthesis and enzymatic ligation may reduce costs by 10- to 100-fold.
For magnetic nanoparticles, scalable coprecipitation and thermal decomposition methods exist but produce particles with size distributions that require additional fractionation. The layer-by-layer assembly used for microflowers is not easily scalable; alternative one-pot synthesis methods are needed.
6. Future Perspectives and Conclusion
The convergence of DNA nanotechnology, synthetic biology, and advanced imaging has created an unprecedented opportunity: the design of autonomous nanorobots that navigate, sense, compute, and treat with precision previously confined to science fiction. The progress reviewed herein — logic-gated DNA origami, magnetic swarm actuation, optoacoustic tracking, and bioresorbable acoustic microrobots — establishes the component technologies. The remaining challenge is integration.
We envision a future "sense-compute-respond" platform with the following specifications: a DNA origami chassis (50–100 nm) decorated with aptamer sensors for pH, MMP-2, and HIF-1α; an AND logic gate implemented via toehold-mediated strand displacement and CHA; a therapeutic payload of doxorubicin or a photosensitizer (e.g., indocyanine green); embedded FePt or Fe₃O₄ nanoparticles for magnetic actuation and optoacoustic contrast; and a PEG coating for immune evasion. Upon intravenous administration, the nanorobot circulates inertly. Upon extravasation into the TME, the low pH and elevated MMP-2 activate the logic gate, triggering drug release. Concurrently, an external magnetic field guides the swarm to the tumor site, and optoacoustic imaging confirms accumulation. Post-treatment, the components degrade or are cleared.
Several near-term advances will accelerate this vision. First, the development of nuclease-resistant DNA chemistries that preserve programmability while extending circulation half-life to hours or days. Second, the demonstration of closed-loop magnetic-optoacoustic guidance in large animal models. Third, the integration of synthetic metabolic pathways — e.g., glucose oxidase-catalase systems — to power actuation from endogenous fuels. Fourth, the validation of logic-gated activation in patient-derived xenograft models to confirm that biomarker thresholds predict therapeutic response.
The field of nanorobotic theranostics stands at an inflection point. The components exist; the design principles are established; the biological barriers are understood. What remains is the engineering — the painstaking integration of subsystems into a cohesive whole, the iterative optimization in animal models, and ultimately, the translation to human patients. The reward, should we succeed, is nothing less than a new paradigm in cancer therapy: precision medicine at the molecular scale, delivered by autonomous agents designed and programmed by human ingenuity.
References
[1] Karna, D., et al. Logic-Gated Modulation of Cell Migration via Mesoscale Mechanical Uncaging Effects. ACS Nano (2025). DOI: 10.1021/acsnano.4c16382.
[2] (CRISPR-Cas13 biosensor study) Computational Design of a CRISPR-Cas13-Driven Genetic Logic Gate Biosensor for Early Detection of Breast Cancer Biomarkers. University of Ruhuna Research Archive (2025).
[3] (Magnetic microbots with optoacoustic imaging) Optoacoustic‐Guided Magnetic Microrobot Platform for Precision Drug Delivery. Advanced Materials (2025). DOI: 10.1002/adma.202511870.
[4] Kim, N., Han, S.Y., Rheem, H.B., et al. Autonomous chemo-metabolic construction of anisotropic cell-in-shell nanobiohybrids in enzyme-powered cell microrobots. Science Advances 11(26), eadu5451 (2025). DOI: 10.1126/sciadv.adu5451.
[5] Aziz, A., Nauber, R., Sánchez Iglesias, A., et al. Nanomaterial-decorated micromotors for enhanced photoacoustic imaging. Journal of Micro and Bio Robotics 19, 37–45 (2023). DOI: 10.1007/s12213-023-00155-8.
[6] Ling, P., Xu, W., Qiao, Y., et al. DNA logic gate for cancer cells identification, detection, and modulation of cell–cell interactions. Microchemical Journal 207, 111840 (2024). DOI: 10.1016/j.microc.2024.111840.
[7] (Continuous input DNA nanomachine) Continuous input DNA Nanomachine to program automated tumor diagnosis and treatment system. Chemical Engineering Journal 495, 153727 (2024).
[8] Han, et al. Imaging-guided bioresorbable acoustic hydrogel microrobots. Science (2024). DOI: 10.1126/science.adn7460.
[9] Nozdriukhin, D., Lyu, S., Bonvin, J., et al. Multifunctional Microflowers for Precise Optoacoustic Localization and Intravascular Magnetic Actuation In Vivo. Advanced Healthcare Materials 14 (2025). DOI: 10.1002/adhm.202403165.


