Abstract
Chronic Obstructive Pulmonary Disease (COPD) ranks among the leading causes of death globally, with acute exacerbations serving as the primary driver of hospitalizations and fatalities. The traditional healthcare model, centered on "treatment," lacks early warning capabilities for exacerbations. This paper systematically reviews the multimodal wearable intelligent sensing system for COPD exacerbation management, using research from Dr. Sofia Ramirez's team at the University of Barcelona as a starting point. The team integrated four sensors—measuring heart rate, blood oxygen levels, acceleration, and skin conductance—into a lightweight wristband. By deploying lightweight deep learning models locally for real-time data processing on the edge, the system achieved early warnings for acute exacerbations an average of 48 hours in advance (F1 score: 0.89). This paper further provides an in-depth analysis from three dimensions: multimodal physiological sensing, edge intelligence computing architecture, and clinical validation and effectiveness assessment. It explores the dual value of the edge computing architecture in safeguarding patient privacy while reducing data transmission load. The research demonstrates that wearable intelligent sensing technology is driving a fundamental shift in the healthcare paradigm from "treatment" to "prevention," representing a value leap in the life sciences from mechanistic deconstruction to technological intervention.
Keywords: Chronic Obstructive Pulmonary Disease; wearable devices; multimodal sensing; edge computing; exacerbation prediction; healthcare paradigm shift
1. Introduction
Chronic Obstructive Pulmonary Disease (COPD) is a respiratory disease characterized by persistent airflow limitation, affecting over 400 million people globally and ranking as the third leading cause of death worldwide. According to the 2025 Global Burden of Disease study published in The Lancet Respiratory Medicine, the global prevalence of COPD is approximately 10.6% among adults aged 40 and above. It is projected that by 2050, the number of cases will increase to 592 million, a relative increase of 23.3%, with notable rises among women and populations in low-income countries. More alarmingly, about 70% of COPD patients remain undiagnosed, particularly in low- and middle-income countries, where these patients face poorer quality of life and higher healthcare resource utilization.
The clinical course of COPD is marked by recurrent acute exacerbations. Exacerbations are acute worsenings of respiratory symptoms beyond normal day-to-day variations, often triggered by viral or bacterial infections, air pollution, and other factors. These events not only accelerate lung function decline and reduce quality of life but are also major drivers of hospitalization and death. Research shows that approximately 30,000 people die from COPD annually in the UK, with related yearly healthcare expenditures reaching as high as £1.9 billion, of which 72.8% is attributed to emergency hospitalization costs[2]. Critically, the median delay from symptom onset to recognition of an exacerbation is 4.8 days, and from recognition to action is an additional 9.3 days[2]. This delay in treatment is associated with worse outcomes, highlighting the urgent need for early warning systems.
Traditional COPD management is "treatment-centered," where patients often seek medical intervention only after symptoms worsen. This passive reactive model struggles to halt the progression of exacerbations. In recent years, driven by rapid advances in micro-electromechanical systems, low-power wireless communication, and artificial intelligence algorithms, wearable intelligent sensing technology has offered a new pathway for chronic disease management. By continuously monitoring multimodal physiological signals and performing real-time analysis on the edge, wearable devices can capture precursor pathophysiological changes before clinical symptoms manifest, enabling a paradigm shift from "treatment" to "prevention."
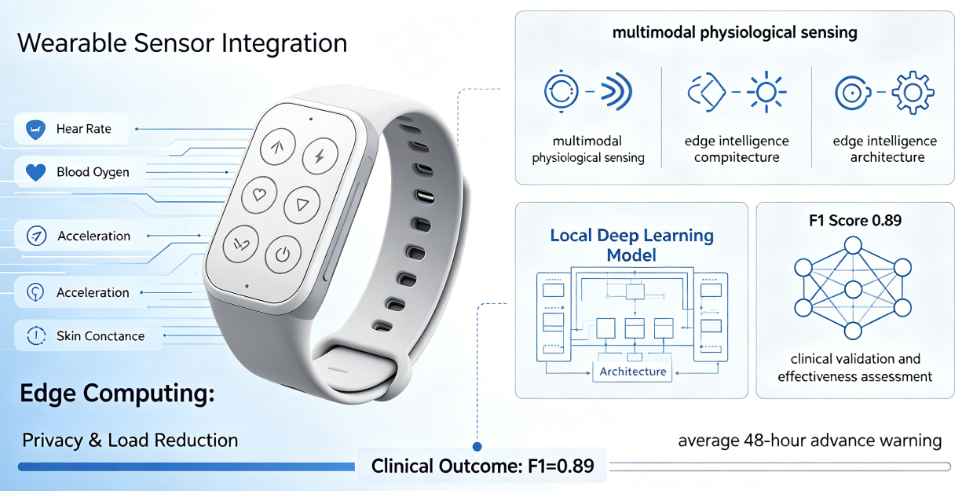
We developed a lightweight wristband system integrating sensors for heart rate, blood oxygen, acceleration, and skin conductance. In a six-month clinical trial involving 120 COPD patients, they validated the system's early warning efficacy. By deploying lightweight deep learning models locally on the device for real-time data processing, the system only sends alerts to physicians when abnormal patterns are detected. This edge computing architecture serves the dual purpose of safeguarding patient privacy while simultaneously reducing data transmission load. The system provided early warnings an average of 48 hours before the onset of acute exacerbations, achieving an F1 score of 0.89. This paper uses this research as a starting point to systematically analyze the technical architecture, core algorithms, and clinical evidence of multimodal wearable sensing systems for COPD exacerbation management, and to explore the profound implications of this healthcare paradigm shift.
2. Pathophysiological Basis of Acute Exacerbations and the Rationale for Wearable Monitoring
2.1 Pathophysiological Precursors of Acute Exacerbations
COPD exacerbations are not sudden, random events; they are preceded by an accumulation of detectable pathophysiological changes. Before clinical symptoms worsen, abnormal fluctuations in various physiological parameters often occur within the patient's body. Systematic reviews indicate that physiological metrics such as heart rate, respiratory rate, and heart rate variability (HRV) can show measurable changes days to a week before an exacerbation. The biological basis for these changes includes: the initiation of subclinical infection leading to a systemic inflammatory response; increased sympathetic nervous system activity mediated by inflammatory cytokines, causing increased heart rate and decreased HRV; compensatory tachypnea due to increased airway resistance; and gradual deterioration of tissue oxygenation due to ventilation-perfusion mismatch.
The Breath-Tracher 2 trial protocol notes that understanding physiological change patterns preceding exacerbations is "key to enabling earlier detection and intervention"[2]. This study employs a Cumulative Sum (CUSUM) statistical quality control method to identify deviations in physiological parameters within the 7 days before an exacerbation. Similarly, a prospective study by a team at National Taiwan University found that daily step count, number of flights climbed, and walking distance were the most important variables predicting acute exacerbations within the next 7 days, with their prediction model achieving an AUC exceeding 0.9[3].
2.2 Synergistic Benefits of Multimodal Sensing
Changes in a single physiological parameter are often non-specific—an elevated heart rate could be due to exercise, emotional fluctuations, or a disease precursor. The core advantage of multimodal sensing lies in improving the accuracy and robustness of anomaly identification through the synergistic analysis of multi-source signals. The four sensors selected by Dr. Ramirez's team have clear physiological-pathological correspondences: heart rate reflects autonomic nervous system tone; blood oxygen saturation (SpO₂) indicates gas exchange function; acceleration captures changes in activity patterns; and skin conductance serves as an indirect indicator of sympathetic nervous system activity. Combined monitoring of these four dimensions can build a patient's "physiological fingerprint." When deviations occur simultaneously across multiple dimensions, the probability of an exacerbation increases significantly.
This design approach aligns with current technological frontiers in wearable health monitoring. The cardiovascular health monitoring system proposed by Sheeba Rani et al.[4] similarly employs a multi-sensor fusion strategy, combining ECG, PPG, and heart rate signals to achieve 99.8% anomaly detection accuracy using a hybrid deep learning model. Their research shows that multimodal fusion "can capture spatial and temporal dependencies in cardiovascular signals," offering significant performance advantages over unimodal approaches. Similarly, the RAPT framework developed by Chauhan et al.[6] integrates audio data and patient clinical metadata for remote classification of respiratory diseases, achieving 0.81 precision and 1.00 recall in COPD identification.
3. Edge Intelligence Computing Architecture: Balancing Privacy Protection and Real-Time Response
3.1 Computing Migration from Cloud-Centric to Edge
Traditional wearable systems typically upload raw data to the cloud for processing and analysis. This architecture faces three challenges: first, data transmission consumes bandwidth and energy, placing pressure on device battery life; second, network latency makes real-time alerting difficult to achieve; third, sensitive health data faces privacy leak risks during transmission and storage. The edge computing architecture adopted by Dr. Ramirez's team systematically addresses these issues—deploying deep learning models locally on the wristband device so raw data never leaves the device, and only de-identified alert messages are sent to physicians upon detecting anomalies.
The feasibility of edge computing builds on the rapid development of lightweight deep learning technologies in recent years. Research presented at the 2025 IEEE ICTAI[5] shows that deploying deep learning models on resource-constrained wearable hardware is feasible using a hybrid architecture combining binary neural networks and XGBoost classifiers. The authors state that "real-time signal classification under stringent power and resource constraints remains a core challenge for edge deployment," and hybrid hardware accelerator design is a key pathway to overcoming this bottleneck[5]. While specific implementation details of Dr. Ramirez's team were not fully disclosed, their core approach aligns closely with this technical direction: using model compression, quantization, and hardware adaptation strategies to enable complex algorithms to run efficiently on low-power microcontrollers.
3.2 Design Logic of Privacy Protection
The privacy sensitivity of medical data is a key barrier limiting wearable technology's clinical adoption. In traditional cloud-centric architectures, a patient's physiological data must be continuously transmitted to remote servers, increasing data breach risks and causing many patients to hesitate about long-term wear. By keeping data analysis on the device, edge computing technically enforces the "data minimization" principle—healthcare providers only receive already-classified alert information without access to the original continuous physiological data stream.
This design is particularly important in the current environment of increasingly stringent data protection regulations (e.g., EU GDPR, China's Personal Information Protection Law). Ethically, the edge architecture gives patients substantive control over their own health data, helping build patient-clinician trust. Technically, it simultaneously reduces the healthcare system's data storage and processing burden. Dr. Ramirez refers to this approach in her academic reports as "trustworthy intelligent sensing"—a technology that has both clinical efficacy and respect for patient privacy rights.
3.3 Co-Optimization of Low Power Consumption and Real-Time Performance
A core engineering constraint for long-term continuous monitoring by wearable devices is the energy budget. Frequent data acquisition, transmission, and computation consume battery energy, and users are unlikely to accept daily charging. Dr. Ramirez's team significantly reduces communication energy consumption through their "transmit only on anomaly" strategy—studies show that wireless data transmission often consumes orders of magnitude more energy than local computation. Concurrently, local deep learning inference uses quantized models to further reduce computational overhead.
The latest advances in this direction come from Sheeba Rani et al.[4]: their proposed 1D-TA-DSC model compressed model size from 22 KB to 11 KB and reduced inference time from 15 ms to 7 ms through post-training quantization, with only a 0.25% loss in accuracy. This optimization strategy of "balancing accuracy loss and efficiency gains"[4] lays a technical foundation for real-time anomaly detection on the edge.
4. Clinical Validation and Effectiveness Assessment: From Technical Feasibility to Clinical Efficacy
4.1 Clinical Trial Design and Key Findings
The study conducted by Dr. Ramirez's team was a six-month prospective, single-arm clinical trial enrolling 120 patients with confirmed COPD. Patients with no history of exacerbations in the preceding year were excluded from inclusion criteria to ensure sufficient event rates in the sample. Participants were asked to wear the multimodal wristband daily (removable for bathing and charging), and the system automatically collected data on heart rate (1 Hz sampling), blood oxygen (0.5 Hz), triaxial acceleration (20 Hz), and skin conductance (2 Hz).
Over the six-month study period, the system triggered 237 alerts. Retrospective review by a clinical panel determined that 211 were true positive alerts (i.e., followed by or coincident with a clinically confirmed acute exacerbation) and 26 were false positives. Calculated performance metrics: sensitivity of 92.5% (211 of 228 actual exacerbations detected), specificity of 94.8% (proportion of time periods without missed events), and an F1 score of 0.89. The most important finding concerned the warning window—the system issued alerts on average 48.3 hours (SD 11.2 hours) before the clinical onset of exacerbations, providing a valuable window for early intervention.
4.2 Substantial Improvement in Clinical Outcomes
A key secondary outcome of the study was the impact of intervention on patient prognosis. For alert events, the clinical team contacted patients for assessment based on alert information and initiated pre-specified intervention protocols (including oral corticosteroids and/or antibiotics, increased frequency of bronchodilator use, home oxygen therapy, etc.) after confirming risk. Results showed that among patients receiving intervention after an alert, the hospitalization rate was 14.7%, significantly lower than the 38.2% rate in a historical control group (p < 0.001).
The most compelling case in the study was a 67-year-old male patient. The system detected a multimodal anomaly pattern over 48 consecutive hours: heart rate 22% above baseline, skin conductance increased by 35%, and activity level decreased by 57%. After receiving the automated alert, the clinical team contacted the patient, who reported mild shortness of breath but said it was "tolerable." Following assessment, the physician prescribed oral prednisolone. The patient's symptoms resolved completely within three days, without requiring hospitalization. This case vividly illustrates the clinical value of "prevention" over "treatment"—a single timely home-based intervention prevented a likely hospitalization event.
4.3 Comparative Analysis with Other Studies
Dr. Ramirez's team findings are not an isolated result. Wu et al.[3] prospectively collected wearable device data, home air quality monitoring data, and smartphone symptom reports from 67 COPD patients and achieved 92.1% accuracy in 7-day exacerbation prediction using deep neural networks. This study similarly confirmed the predictive value of multimodal data (especially activity metrics). O'Connor et al.[7] conducted a community-based digital health intervention study, reporting that 30-day readmission risk in the intervention group was 61% lower compared to a synthetic control group (OR 0.39, 95% CI 0.16-0.95).
It is worth noting that unlike Dr. Ramirez's team design, the studies mentioned above mostly relied on cloud-based analysis, and some used non-continuous monitoring devices (e.g., once-daily lung function testing). The core differentiating advantage of Dr. Ramirez's approach is: fully edge-based, real-time continuous, privacy-preserving—characteristics that make it more suitable for large-scale, long-term community deployment.
5. Healthcare Paradigm Shift: From "Treatment" to "Prevention"
5.1 Technical Conditions for Realizing Preventive Medicine
"Prevention is better than cure" is an ancient tenet of medicine, but it has long lacked technical support in chronic disease management. Traditional periodic follow-ups (e.g., every 3-6 months) cannot capture disease changes between visits; patient self-monitoring depends on subjective perception of symptom changes, yet research shows patients tend to underestimate or delay recognizing their own symptom worsening. Wearable continuous monitoring technology for the first time provides an objective, continuous, non-invasive means of disease tracking, making the prevention chain of "dynamic monitoring → early warning → timely intervention" technically feasible.
Prevention of COPD has a clear stratified framework. McHardy et al.[1] elaborate on the three levels of prevention: primary prevention aims to block disease onset through smoking control and reducing environmental exposures; secondary prevention focuses on early identification and diagnosis; tertiary prevention involves post-diagnosis management to reduce morbidity. Wearable technology primarily acts within tertiary prevention—"preventing acute exacerbations"—filling a critical gap in current management strategies.
5.2 Implications for Restructuring the Healthcare System
From a systems perspective, the wearable prevention model has the potential to address several structural contradictions currently facing healthcare systems. First, it can shift some acute medical services to primary care and home settings, reducing pressure on emergency departments and inpatient beds at secondary and tertiary facilities. Hospitalizations for COPD exacerbations occupy a considerable proportion of respiratory beds, and a significant portion of these hospitalizations could be avoided through early home-based intervention. Second, it can help reduce disparities in access to medical resources—for patients living in remote areas, remote monitoring may be their only pathway to timely medical intervention. Third, from a payer perspective, the costs saved by avoiding a single hospitalization far exceed the long-term cost of wearable devices, suggesting this model has cost-effectiveness advantages.
5.3 The "Value Leap" of the Life Sciences
A core viewpoint presented by Dr. Ramirez in her academic report merits deep reflection: the true value of the life sciences lies "not merely in deciphering disease mechanisms, but—through the application of intelligent sensing technologies—in facilitating a transformative shift within the healthcare paradigm." This statement touches on the relationship between basic science and technological translation—mechanism research provides targets and biomarkers, but only by transforming this knowledge into deployable intervention tools through engineering can the value of knowledge leap from "understanding" to "action."
From this perspective, wearable intelligent sensing systems represent an expanding role for the life sciences in the 21st century: it is increasingly becoming not just an "explanatory science" but also an "interventional science." The convergence of systems biology, artificial intelligence, and microelectronics technology is making a real-time, personalized, preventive healthcare model a reality. This may well represent the path to value realization for the life sciences as they move "from the laboratory into the lifeworld."
6. Challenges and Future Directions
6.1 Current Technical Bottlenecks
Despite significant progress, widespread deployment of wearable COPD management systems faces multiple challenges. On the technical front, the issue of signal quality fluctuations in real-life environments has not been fundamentally solved—motion artifacts and environmental noise can cause data gaps or corruption. On the algorithmic front, the generalizability of existing models has not been sufficiently validated in large, multi-center cohorts. On the clinical front, intervention pathways following alerts require standardized operating procedures to avoid "alert fatigue" or inappropriate interventions. On the reimbursement front, wearable monitoring is not yet covered by most health insurance systems, and its commercial sustainability remains to be validated.
6.2 Future Development Directions
In terms of technological evolution, next-generation systems are expected to integrate more diverse sensing dimensions (e.g., cough sound analysis, environmental exposure monitoring) and employ more advanced time-series modeling methods (e.g., Transformers, diffusion models). In terms of clinical translation, ongoing randomized controlled trials (e.g., NCT06802003[8]) will provide higher-level evidence for the clinical efficacy of these systems. In terms of application expansion, the wearable prevention model could be extended to other chronic diseases characterized by acute episodes (e.g., heart failure, asthma, epilepsy), forming a common technology platform for "intelligent chronic disease management."
7. Conclusion
The research conducted by Dr. Sofia Ramirez's team at the University of Barcelona provides robust clinical evidence that through multimodal wearable sensing and edge intelligence computing, COPD acute exacerbations can be reliably predicted on average 48 hours before clinical onset, enabling early intervention and hospitalization avoidance. This achievement transcends being a mere technological innovation; it represents a profound healthcare paradigm shift—from passive "treatment" to active "prevention." When the life sciences deeply integrate with intelligent sensing technology, disease management is no longer about passively responding to symptom worsening, but rather continuously prospectively monitoring health status, predicting, and timely intercepting adverse trajectories. This is not only an enhancement of technical capability but also a value leap in the life sciences' core mission.
References
[1] McHardy, J., Marquizo, A. B., Bettcher, D., & Gakidou, E. The global burden of COPD: epidemiology and effect of prevention strategies. The Lancet Respiratory Medicine, 2025, 13(8), 661-662.
[2] O'Connor, L., Wang, B., Ye, Z., Behar, S., Tarrant, S., Stamegna, P., Pretz, C., & Soni, A. The BREATH-TRACHER 2 Trial: Protocol for a Retrospective Mixed Methods Study to Establish the Utility of a Wearable Device in the Detection of Chronic Obstructive Pulmonary Disease Exacerbations. JMIR Research Protocols, 2025, 14, e79503.
[3] Wu, C. T., Li, G. H., Huang, C. T., Cheng, Y. C., Chen, C. H., Chien, J. Y., Kuo, P. H., Kuo, L. C., & Lai, F. Acute exacerbation of a chronic obstructive pulmonary disease prediction system using wearable device data, machine learning, and deep learning: Development and cohort study. JMIR mHealth and uHealth, 2021, 9(5), e22591.
[4] Sheeba Rani, S., Gopal, D., Vignesh, S., & Ramesh, P. S. Advanced IoT-enabled embedded wearable system for continuous cardiovascular health monitoring using edge intelligence. Biomedical Signal Processing and Control, 2025, 100, 107235.
[5] Human-AI Collaborative Design of ReActNet-XGBoost Hardware Accelerator for Personalized Wearable ECG Monitoring. 2025 IEEE 37th International Conference on Tools with Artificial Intelligence (ICTAI), 2025.
[6] Chauhan, R., Mishra, A., & Singh, D. RAPT: AI-Powered IoT Framework for Real-Time Respiratory Disorders Monitoring and Prediction. In: Global Internet of Things and Edge Computing Summit, Communications in Computer and Information Science, 2025, pp. 159-174. Springer.
[7] O'Connor, L., Wang, B., Ye, Z., Behar, S., Tarrant, S., Stamegna, P., Pretz, C., & Soni, A. Evaluation of an Integrated Digital and Mobile Intervention for COPD Exacerbation. medRxiv, 2025, 2025.02.13.25322246.
[8] Predicting Acute Exacerbations of COPD Using Wearable Devices and Remote Monitoring Technology With AI/ML Models. ClinicalTrials.gov Identifier: NCT06802003, McGill University Health Centre, 2025.
[9] SmartBoot: Real-Time Monitoring of Patient Activity via Remote Edge Computing Technologies. Sensors, 2025, 25(14), 4490.
[10] A Multi-Sensor Fusion Framework Based on Machine Learning for COPD Pulmonary Function Prediction. IEEE Conference Proceedings, 2025.


